Pyrolysis-GC/MS
This introduction assumes the reader has skills and experiences in GC and GC/MS analysis or, having no or limited experiences in pyrolysis-GC (Py-GC), is willing to learn and use it in the future.
2. Pyrolysis
2.1 What is pyrolysis?
Pyrolysis is the decomposition of a polymer into smaller molecules by heating at high temperatures. In most cases, this is done under inert atmosphere (no oxygen). Fission of molecular bonds and physical changes irreversibly take place. These smaller molecules obtained by fission in an inert atmosphere are called Pyrolyzates.
Analytical pyrolysis using a Pyrolysis (Py)-GC/MS system is described.

A polymer is made up of repetitive atoms or molecules each covalently bonded, forming a long molecular chain or macromolecule.
In analytical pyrolysis, these molecular chains of a polymer are broken up by rapidly heating to a high temperature usually at about 600 ºC, and the composition of the polymer is investigated by introducing the smaller molecules (pyrolyzates) into an analytical instrument such as GC/MS.
(An illustrative image is shown in the figure on the left.)
2.2 Pyrolysis and pyrolyzates of polymeric materials
Hydrocarbon, C4 butane in this example, burns to give carbon dioxide and water when heated to high temperatures in air or in the presence of oxygen.
On the other hand, under non-oxygen atmosphere, C-C bond scissions occurs to give smaller molecules.

In analytical pyrolysis, polymers with large molecular weight are heated at high temperatures under the non-oxygen atmosphere, affording thermally decomposed products including monomers. The molecular structure of polymers can be elucidated from the information.

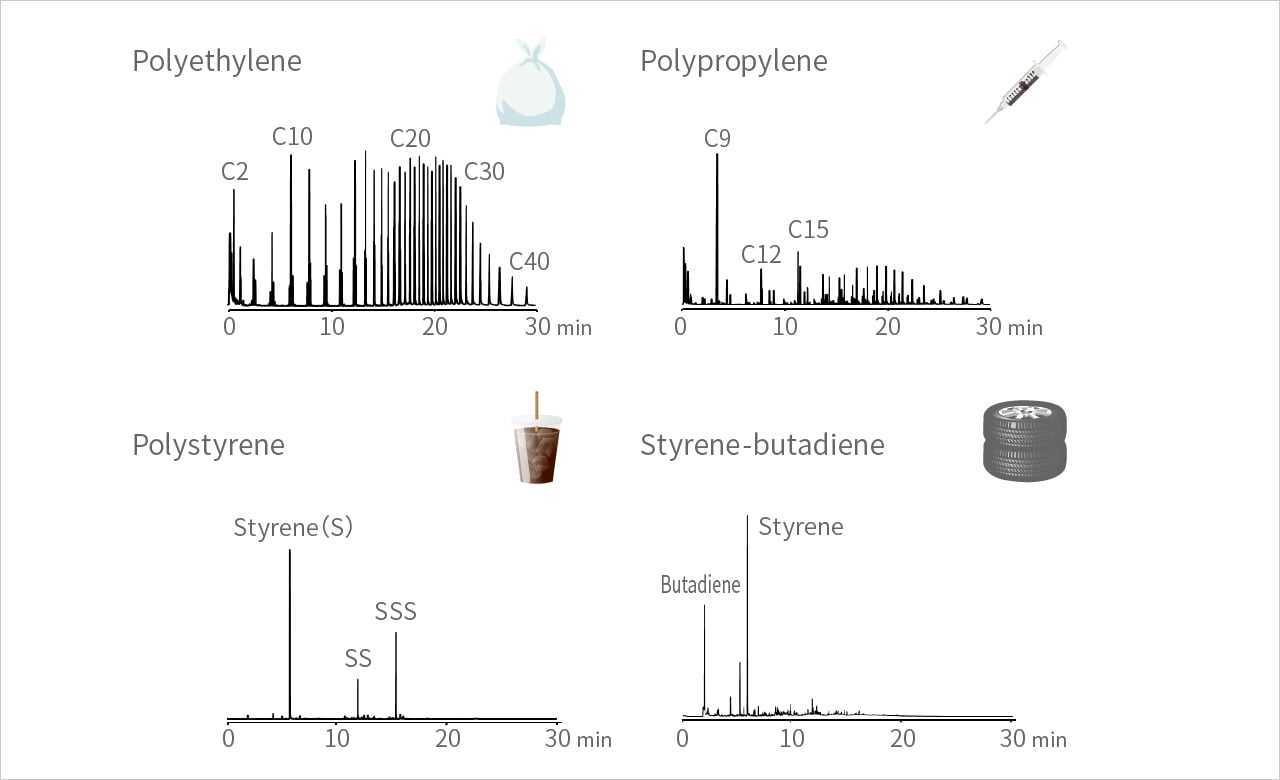
2.3 Pyrograms of typical polymers
A pyrogram is a chromatogram of the pyrolyzates obtained by pyrolysis.
Shown below are pyrograms obtained by flash pyrolysis of typical polymers at 600 ºC under helium (He) atmosphere. The profile of a pyrogram differs greatly from polymer to polymer.
Each peak on the pyrogram represents a smaller molecule (pyrolyzate) derived from the decomposition of a polymer. Therefore, in most cases, identifying each peak on the pyrogram can lead to the identification of the original polymer.
Polyethylene pyrolysis produces hydrocarbon fragments (pyrolyzates) from C2 to C100 or more, in a characteristic way, thus it is easy to determine the original structure from the pyrolyzates.
Polypropylene and polystyrene are pyrolyzed to give dimer, trimer, and others, in addition to monomer. Copolymers decompose to give even more complex profiles.